Live webcast
Technical Documentation
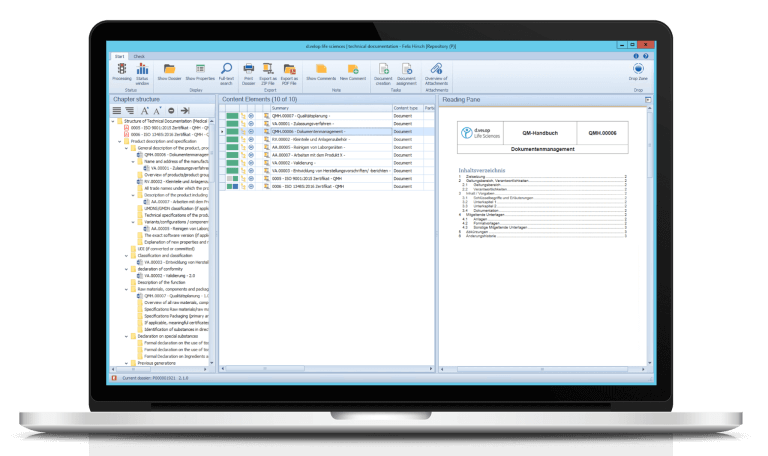
Support and simplification of technical documentation
- Learn how our module “dls | technical documentation” supports and simplifies the creation and updating of your technical documentation.
- Find out to what extent you will be able to digitally implement the requirements of the Medical Device Regulation (MDR) in the future.
- Find out how versatile our process-oriented dossier management can be, e.g. for the creation of site master files, machine and plant dossiers, building dossiers, QM documentation, project folders, and much more.
What you can expect
1. Presentation of Digital Life Sciences GmbH
2. Overview of solutions from Digital Life Sciences GmbH
3. Live presentation of a sample use case
4. Summary
5. References
Just a few steps to your goal:
1. Please use the form “Request Live Webcast” below
2. Fill in the contact fields.
3. We will approach you. By telephone or e‑mail.
Note on the live webcast: Specify your preferred date with one week’s notice.
Request live webcast
Request live webcast
Just a few steps to your goal:
1. Use the form below and select your preferred date.
2. Select a time and enter your contact details.
3. You will automatically receive an e-mail with a Teams meeting and further information.
Your contact person: Georg Langbehn

Your WebCast consultant
Georg Langbehn works as a Product Owner at Digital Life Sciences and looks after a large number of new and existing customers. In this role, he has specalized on the presentation and consulting of Technical Documentation and other related topics.
